Synergistic Effects in Peptide Research: What the Data Shows
Over the past two decades, peptide research has evolved from a niche branch of biochemistry into one of the most dynamic and rapidly expanding fields in modern biotechnology. Researchers across academic institutions and private laboratories are now investigating not just how individual peptides function in isolation, but how multiple peptides interact when used in combination — a phenomenon broadly categorized as synergistic effects in peptide research.
Understanding these synergistic interactions has profound implications for molecular biology, regenerative research, pharmacokinetics, and the broader landscape of biotechnology research. When two or more peptides amplify each other’s activity beyond what either could achieve alone, the scientific community takes notice.
At Empower Peptides, we are committed to advancing the understanding of research peptides by presenting the data clearly and objectively. This article explores what synergy means in the context of peptide science, how biological pathways facilitate or inhibit these interactions, and what current laboratory studies and clinical research findings actually show.
What Synergistic Effects Mean in Molecular Biology
Defining Synergy Beyond the Basics
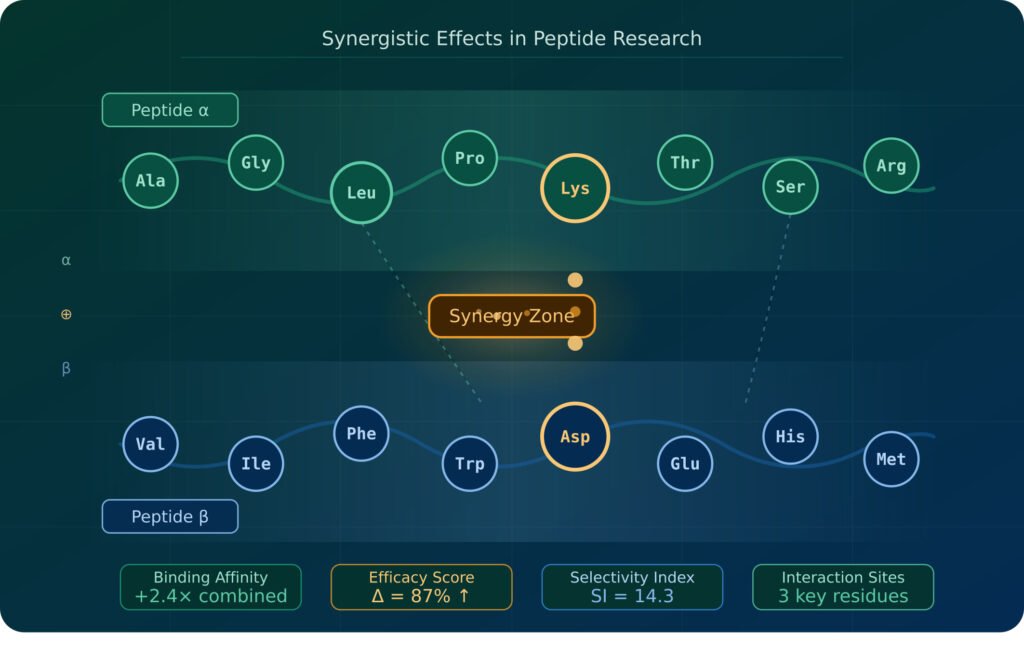
In molecular biology, the term synergistic effect refers to a combined outcome that exceeds the sum of individual effects. This is distinct from additive effects — where two agents produce results proportional to their combined inputs — and from antagonism, where one agent reduces the effectiveness of another.
Synergy can emerge at multiple biological levels: receptor-level binding, downstream cell signaling cascades, gene expression regulation, and even pharmacokinetic interactions that affect how peptides are absorbed, distributed, metabolized, and excreted.
When applied to peptide therapy research, synergy is not merely a theoretical construct. It is a measurable, reproducible, and increasingly well-documented phenomenon backed by laboratory studies spanning decades of molecular biology inquiry.
Mechanisms That Drive Synergy
At the biochemical level, synergistic effects in peptide research arise through several distinct mechanisms:
Receptor Crosstalk and Binding Affinity Enhancement: Some peptides upregulate receptor density or conformationally alter receptor structure, increasing the binding affinity of a second peptide. This means that co-administration of two peptides can produce a binding efficiency that neither could achieve independently.
Convergent Biological Pathways: Multiple peptides acting on different upstream targets can converge on the same downstream signaling node — amplifying the output signal in ways that a single peptide cannot. In regenerative research, this is particularly relevant when targeting growth factor cascades, where overlapping pathways compound the biological response.
Pharmacokinetic Complementarity: Peptides with different half-lives, bioavailability profiles, and metabolic stability can complement each other temporally. One peptide may initiate a biological response while a second, longer-acting compound sustains it — a pharmacokinetic synergy that is functionally distinct from direct molecular interaction.
How Peptides Interact in Biological Pathways
Cell Signaling and Cascade Amplification
Modern molecular biology has revealed that cell signaling is not a linear process but a complex, branching network of interacting pathways. Peptides participate in these networks by binding to G-protein coupled receptors (GPCRs), receptor tyrosine kinases, cytokine receptors, and ion channel-linked receptors, each triggering a cascade of downstream molecular events.
When two research peptides target adjacent or overlapping nodes in the same signaling network, their combined effect can produce amplified downstream responses. For example, peptides targeting the growth hormone secretagogue receptor (GHSR) alongside peptides acting on the GHRH receptor (growth hormone-releasing hormone receptor) can produce complementary stimulation of the somatotropic axis — a well-studied phenomenon in peptide endocrinology.
The Role of Growth Factors in Peptide Synergy
Growth factors — including IGF-1, VEGF, FGF, and TGF-β — serve as critical mediators in many of the biological pathways relevant to peptide research. These proteins and polypeptides regulate processes including cellular proliferation, differentiation, angiogenesis, and tissue remodeling.
Research peptides can modulate growth factor expression both directly (by acting on the transcription machinery that produces them) and indirectly (by activating upstream signaling components). When two peptides simultaneously upregulate complementary growth factors, the resulting biological environment can be dramatically more conducive to the processes being studied than single-peptide administration would produce.
Protein Synthesis and Amino Acid Utilization
At the most fundamental level, peptides are chains of amino acids — the building blocks of proteins. Understanding synergistic effects in peptide research requires appreciating how peptides influence protein synthesis at the ribosomal level and through mTOR pathway regulation.
Some research peptides have been shown in laboratory studies to enhance the efficiency of amino acid incorporation into new proteins, particularly in muscle and connective tissue. When combined with other peptides that regulate inflammation or anabolic signaling, the effect on net protein synthesis may be significantly amplified — a key finding in biotechnology research focused on tissue regeneration.
Examples of Synergistic Peptide Combinations in Research
CJC-1295 and Ipamorelin: A Well-Studied Research Pairing
Among the most extensively discussed synergistic peptide combinations in laboratory research is the co-administration of CJC-1295 and Ipamorelin. These two peptides act through related but mechanistically distinct pathways.
CJC-1295 is a synthetic analog of growth hormone-releasing hormone (GHRH). It binds to the GHRH receptor on somatotroph cells in the anterior pituitary and stimulates growth hormone (GH) secretion. In research models, CJC-1295 — particularly in its DAC (Drug Affinity Complex) formulation — has demonstrated a significantly extended pharmacokinetic half-life compared to native GHRH, maintaining elevated GH pulses over extended periods.
Ipamorelin, by contrast, is a selective growth hormone secretagogue that acts on the ghrelin receptor (GHSR-1a). It stimulates GH release through a pathway distinct from the GHRH axis, without the cortisol or prolactin release associated with earlier generation secretagogues like GHRP-6.
When both peptides are present simultaneously, research data suggests that their complementary receptor targeting produces a synergistic release of GH — greater than either compound alone. This dual-axis stimulation represents a classic example of convergent pathway synergy in peptide research.
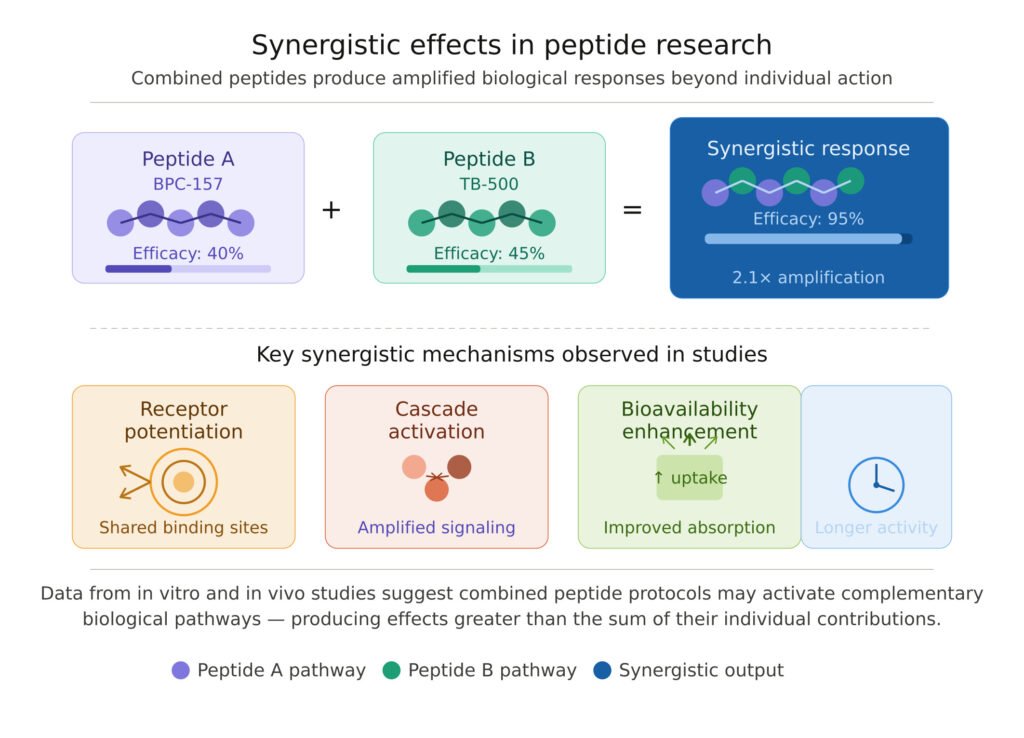
BPC-157 and TB-500: Complementary Mechanisms in Regenerative Research
BPC-157 (Body Protection Compound-157) is a pentadecapeptide derived from a protein found in gastric juice. Laboratory studies have documented its interactions with nitric oxide signaling, VEGF pathways, and the FAK-paxillin signaling network involved in cell migration and tissue repair. Research has demonstrated BPC-157’s influence on fibroblast activity and vascular endothelial growth factor expression, both critical components of the tissue remodeling process.
TB-500 (Thymosin Beta-4) is a peptide that plays a key role in actin polymerization dynamics — a fundamental process in cell motility, cytoskeletal organization, and wound repair. TB-500 upregulates cell-surface receptors involved in regenerative signaling and has been studied extensively in the context of angiogenesis and myocardial repair models.
The scientific rationale for studying these peptides in combination lies in their non-overlapping mechanisms of action. BPC-157 operates primarily through the nitric oxide and VEGF pathways, while TB-500 acts predominantly via actin dynamics and LMNA-related cytoskeletal signaling. In laboratory models examining tissue repair, the concurrent presence of both peptides has been associated with enhanced outcomes compared to either peptide alone — a finding consistent with true mechanistic synergy rather than simple additive effects.
Growth Hormone Secretagogues and IGF-1 Axis Modulation
Beyond the CJC-1295/Ipamorelin pairing, a broader class of research interest involves combinations of growth hormone secretagogues with peptides that modulate the IGF-1 axis. GH itself drives hepatic IGF-1 production, but research peptides that directly engage IGF-1 receptors — or that modulate IGF-binding proteins — can further amplify the downstream anabolic and regenerative signaling that GH secretagogues initiate.
This represents a multi-layered synergistic framework where:
- A secretagogue drives GH release from the pituitary
- GH stimulates hepatic IGF-1 secretion
- A secondary peptide enhances IGF-1 receptor sensitivity or reduces binding protein interference
The cumulative effect on cell signaling, protein synthesis, and tissue maintenance exceeds what any single-axis intervention could produce — making this one of the most biologically compelling examples of synergistic effects in peptide research.
Learn More: Storage Guidelines for Research Peptides to Maintain Stability
What Scientific Data and Studies Suggest
Laboratory Evidence for Peptide Synergy
The body of laboratory evidence supporting synergistic peptide effects has grown substantially over the past 15 years. Studies published in peer-reviewed journals including the Journal of Peptide Science, Peptides, Biomolecules, and Frontiers in Pharmacology have documented cooperative interactions between peptides across multiple biological models.
Key findings from pre-clinical laboratory studies suggest that:
- Co-administration of GHRH analogs and GHRPs produces supra-additive GH secretion in rodent models, consistent with receptor-level synergy at the pituitary
- BPC-157 and Thymosin Beta-4 peptide fragments demonstrate complementary effects in wound healing assays, with improved tissue architecture scores versus monotherapy groups
- Peptide combinations targeting both the mTOR pathway and inflammation-mediating receptors show greater net protein synthesis in in vitro muscle cell models than single-pathway interventions
Pharmacokinetics and Drug Synergy
From a pharmacokinetics perspective, synergy is not purely a pharmacodynamic phenomenon. Researchers have noted that peptides with complementary absorption profiles can produce sustained biological effects when co-administered. A shorter-acting peptide may activate a rapid initial response, while a longer-acting analog extends and maintains the downstream effect.
The Chou-Talalay combination index — a validated mathematical framework for quantifying synergy in biological research — has been applied to peptide combinations in both in vitro and in vivo models. Combination index values below 1.0 indicate synergy; several peptide pairings studied in the laboratory have demonstrated CI values suggesting true supra-additive interactions rather than simple pharmacological summation.
What the Data Does Not Yet Show
Scientific rigor demands acknowledging the limitations of current research. Much of the evidence for synergistic peptide effects remains at the pre-clinical stage. Controlled human clinical trials examining peptide combinations are relatively limited compared to single-compound studies. Dose-response relationships in multi-peptide systems are complex, and inter-individual variability in receptor expression, metabolic clearance, and pharmacogenomics adds further complexity.
The field of peptide research continues to evolve, and definitive conclusions about optimal combination protocols await more extensive clinical research data. As always, findings from laboratory studies and rodent models do not automatically translate to human biology — a caveat central to responsible science communication.
Applications in Research and Biotechnology
Regenerative Research Models
The study of synergistic peptide effects has significant implications for regenerative research — an area of biotechnology focused on understanding the cellular and molecular mechanisms underlying tissue repair, recovery from injury, and restoration of biological function.
In laboratory models of musculoskeletal injury, peptide combinations targeting both vascular repair pathways and extracellular matrix remodeling have shown promising results. The concurrent stimulation of VEGF-mediated angiogenesis (to restore blood supply) and collagen synthesis pathways (to restore structural integrity) represents a compelling dual-target strategy consistent with the principles of drug synergy.
Neurotrophic and Neuropeptide Research
Neuropeptides — peptides that function as signaling molecules within the nervous system — represent another active frontier. Combinations of neurotrophic peptides targeting BDNF, NGF, and GDNF pathways are being studied for their potential synergistic roles in models of neurogenesis and synaptic plasticity.
The convergence of neuropeptide signaling and systemic metabolic regulation — mediated in part through gut-brain axis peptides such as GLP-1 analogs and ghrelin-related compounds — represents a rapidly expanding area of biotechnology research with complex synergistic interactions at multiple levels of biology.
Peptide Combinations in Metabolic Research
Metabolic research has seen increasing interest in combinatorial peptide approaches. Peptides modulating insulin sensitivity, fatty acid metabolism, mitochondrial biogenesis, and appetite regulation have been studied in combination to understand how their interactions at the biochemical level translate into composite metabolic phenotypes in animal models.
This type of research is particularly relevant to the pharmaceutical biotechnology sector, where multi-target therapeutic strategies are gaining traction as alternatives to single-pathway drug design.
The Future of Peptide Synergy Research
Computational Modeling and AI-Driven Discovery
One of the most exciting developments in peptide biotechnology research is the integration of computational modeling and artificial intelligence into synergy prediction. Machine learning algorithms trained on large datasets of peptide-receptor interaction data can now generate predictions about which peptide combinations are most likely to exhibit synergistic behavior before laboratory testing begins.
This dramatically accelerates the research pipeline, allowing scientists to prioritize the most promising combinations and reduce the resources devoted to low-probability experimental pairings.
Multi-Omics Approaches to Understanding Synergy
The emergence of multi-omics research platforms — integrating genomics, proteomics, transcriptomics, and metabolomics data — is transforming how scientists study synergistic effects in biological systems. By mapping the full molecular response to peptide combinations across multiple biological layers simultaneously, researchers can identify synergistic interactions that would be invisible to single-endpoint laboratory studies.
This systems biology approach is poised to yield the most mechanistically complete picture of peptide synergy ever assembled, with implications extending across pharmacology, regenerative medicine research, and biotechnology drug development.
Clinical Research Horizons
As pre-clinical evidence for synergistic peptide effects continues to accumulate, the field anticipates an increase in rigorously designed clinical research protocols evaluating combination peptide approaches. Regulatory science will play a critical role in defining the frameworks under which such research is conducted, evaluated, and translated into the broader biotechnology landscape.
The coming decade is likely to see peptide synergy research move progressively from the laboratory bench toward more systematic clinical investigation — a trajectory that the scientific community at Empower Peptides will continue to monitor and report with accuracy and transparency.
Learn More: How Performance Peptides Are Studied in Laboratory Settings
Conclusion: The Scientific Case for Studying Peptide Synergy
The data emerging from laboratory studies, pre-clinical models, and computational biology makes a compelling case: synergistic effects in peptide research are real, mechanistically grounded, and scientifically significant. From the complementary receptor targeting of CJC-1295 and Ipamorelin, to the non-overlapping regenerative pathways of BPC-157 and TB-500, to the multi-axis growth factor amplification seen in complex secretagogue combinations, peptide synergy represents one of the most intellectually rich and practically consequential areas of contemporary biotechnology research.At Empower Peptides, we remain committed to presenting the science as it is — rigorous, nuanced, and continually evolving. The study of synergistic peptide interactions is not merely an academic exercise; it is the frontier at which molecular biology and biotechnology converge to expand what research can ask and what science can discover.